 |
1 |  | 
What is the relationship between 1-butene and cis-2-butene? |
|  | A) | unrelated compounds |
|  | B) | constitutional isomers |
|  | C) | enantiomers |
|  | D) | diastereomers |
 |
 |
2 |  | 
What is the relationship between trans-2-butene and cis-2-butene? |
|  | A) | unrelated compounds |
|  | B) | constitutional isomers |
|  | C) | enantiomers |
|  | D) | diastereomers |
 |
 |
3 |  | 
Determine the relationship between the two molecules shown.
 <a onClick="window.open('/olcweb/cgi/pluginpop.cgi?it=jpg::::/sites/dl/free/0072397462/209333/5_3_eq.jpg','popWin', 'width=NaN,height=NaN,resizable,scrollbars');" href="#"><img valign="absmiddle" height="16" width="16" border="0" src="/olcweb/styles/shared/linkicons/image.gif"> (16.0K)</a> <a onClick="window.open('/olcweb/cgi/pluginpop.cgi?it=jpg::::/sites/dl/free/0072397462/209333/5_3_eq.jpg','popWin', 'width=NaN,height=NaN,resizable,scrollbars');" href="#"><img valign="absmiddle" height="16" width="16" border="0" src="/olcweb/styles/shared/linkicons/image.gif"> (16.0K)</a> |
|  | A) | constitutional isomers |
|  | B) | enantiomers |
|  | C) | diastereomers |
|  | D) | identical molecules |
 |
 |
4 |  | 
Determine the relationship between the two molecules shown.
 <a onClick="window.open('/olcweb/cgi/pluginpop.cgi?it=jpg::::/sites/dl/free/0072397462/209333/5_4_eq.jpg','popWin', 'width=NaN,height=NaN,resizable,scrollbars');" href="#"><img valign="absmiddle" height="16" width="16" border="0" src="/olcweb/styles/shared/linkicons/image.gif"> (14.0K)</a> <a onClick="window.open('/olcweb/cgi/pluginpop.cgi?it=jpg::::/sites/dl/free/0072397462/209333/5_4_eq.jpg','popWin', 'width=NaN,height=NaN,resizable,scrollbars');" href="#"><img valign="absmiddle" height="16" width="16" border="0" src="/olcweb/styles/shared/linkicons/image.gif"> (14.0K)</a> |
|  | A) | constitutional isomers |
|  | B) | enantiomers |
|  | C) | diastereomers |
|  | D) | identical molecules |
 |
 |
5 |  | 
Give all terms that describe compounds (a) and (b).
 <a onClick="window.open('/olcweb/cgi/pluginpop.cgi?it=jpg::::/sites/dl/free/0072397462/209333/5_5_eq.jpg','popWin', 'width=NaN,height=NaN,resizable,scrollbars');" href="#"><img valign="absmiddle" height="16" width="16" border="0" src="/olcweb/styles/shared/linkicons/image.gif"> (16.0K)</a> <a onClick="window.open('/olcweb/cgi/pluginpop.cgi?it=jpg::::/sites/dl/free/0072397462/209333/5_5_eq.jpg','popWin', 'width=NaN,height=NaN,resizable,scrollbars');" href="#"><img valign="absmiddle" height="16" width="16" border="0" src="/olcweb/styles/shared/linkicons/image.gif"> (16.0K)</a> |
|  | A) | enantiomers |
|  | B) | diastereomers |
|  | C) | same compound |
|  | D) | chiral molecules |
 |
 |
6 |  | 
How many stereoisomers are there for the following structure?
 <a onClick="window.open('/olcweb/cgi/pluginpop.cgi?it=jpg::::/sites/dl/free/0072397462/209333/5_6_eq.jpg','popWin', 'width=NaN,height=NaN,resizable,scrollbars');" href="#"><img valign="absmiddle" height="16" width="16" border="0" src="/olcweb/styles/shared/linkicons/image.gif"> (11.0K)</a> <a onClick="window.open('/olcweb/cgi/pluginpop.cgi?it=jpg::::/sites/dl/free/0072397462/209333/5_6_eq.jpg','popWin', 'width=NaN,height=NaN,resizable,scrollbars');" href="#"><img valign="absmiddle" height="16" width="16" border="0" src="/olcweb/styles/shared/linkicons/image.gif"> (11.0K)</a> |
|  | A) | 1 |
|  | B) | 2 |
|  | C) | 3 |
|  | D) | 4 |
 |
 |
7 |  | 
Which of the following molecules exists as a pair of enantiomers? |
|  | A) | 2-Bromopropane |
|  | B) | 1-Bromo-3-methylbutane |
|  | C) | 2-Cyclohexen-1-ol |
|  | D) | cis-1,2-Dichlorocyclobutane |
 |
 |
8 |  | 
Which of the following diols exists as a pair of enantiomers? |
|  | A) | cis-1,3-Cyclohexanediol |
|  | B) | trans-1.3-Cyclohexanediol |
|  | C) | cis-1,4-Cyclohexanediol |
|  | D) | trans-1,4-Cyclohexanediol |
 |
 |
9 |  | 
Which of the following is capable of existing as a pair of enantiomers? |
|  | A) | 2-methylpropane |
|  | B) | 2-methylpentane |
|  | C) | 3-methylpentane |
|  | D) | 3-methylhexane |
 |
 |
10 |  | 
Which of the 7 isomers of dichlorocyclohexane possess a plane of symmetry? |
|  | A) | cis-1,2; cis-1,3; |
|  | B) | 1,1; cis-1,2; cis-1,3; |
|  | C) | cis-1,2; cis-1,3; cis-1,4; |
|  | D) | 1,1; cis-1,2; cis-1,3; cis-1,4; trans-1,4 |
 |
 |
11 |  | 
Which of the following has a plane of symmetry? |
|  | A) | cis-1,2-Dimethylcyclohexane |
|  | B) | trans-1,3-Dimethylcyclohexane |
|  | C) | 1,3-Dimethylcyclohexene |
|  | D) | 1,4-Dimethylcyclohexene |
 |
 |
12 |  | 
What is the IUPAC name of the following molecule?
 <a onClick="window.open('/olcweb/cgi/pluginpop.cgi?it=jpg::::/sites/dl/free/0072397462/209333/5_12_eq.jpg','popWin', 'width=NaN,height=NaN,resizable,scrollbars');" href="#"><img valign="absmiddle" height="16" width="16" border="0" src="/olcweb/styles/shared/linkicons/image.gif"> (13.0K)</a> <a onClick="window.open('/olcweb/cgi/pluginpop.cgi?it=jpg::::/sites/dl/free/0072397462/209333/5_12_eq.jpg','popWin', 'width=NaN,height=NaN,resizable,scrollbars');" href="#"><img valign="absmiddle" height="16" width="16" border="0" src="/olcweb/styles/shared/linkicons/image.gif"> (13.0K)</a> |
|  | A) | (3R,5S)-dibromohexane |
|  | B) | (2R,4R)-dibromohexane |
|  | C) | (2R,4S)-dibromohexane |
|  | D) | (R,S)-dibromohexane |
 |
 |
13 |  | 
Which of the following groups has the highest priority in the (R,S) system? |
|  | A) |  <a onClick="window.open('/olcweb/cgi/pluginpop.cgi?it=jpg::::/sites/dl/free/0072397462/209333/5_13A.jpg','popWin', 'width=NaN,height=NaN,resizable,scrollbars');" href="#"><img valign="absmiddle" height="16" width="16" border="0" src="/olcweb/styles/shared/linkicons/image.gif"> (10.0K)</a> <a onClick="window.open('/olcweb/cgi/pluginpop.cgi?it=jpg::::/sites/dl/free/0072397462/209333/5_13A.jpg','popWin', 'width=NaN,height=NaN,resizable,scrollbars');" href="#"><img valign="absmiddle" height="16" width="16" border="0" src="/olcweb/styles/shared/linkicons/image.gif"> (10.0K)</a> |
|  | B) | –CH=CH2 |
|  | C) | –CH2–CH3 |
|  | D) |  <a onClick="window.open('/olcweb/cgi/pluginpop.cgi?it=jpg::::/sites/dl/free/0072397462/209333/5_14_D.jpg','popWin', 'width=NaN,height=NaN,resizable,scrollbars');" href="#"><img valign="absmiddle" height="16" width="16" border="0" src="/olcweb/styles/shared/linkicons/image.gif"> (12.0K)</a> <a onClick="window.open('/olcweb/cgi/pluginpop.cgi?it=jpg::::/sites/dl/free/0072397462/209333/5_14_D.jpg','popWin', 'width=NaN,height=NaN,resizable,scrollbars');" href="#"><img valign="absmiddle" height="16" width="16" border="0" src="/olcweb/styles/shared/linkicons/image.gif"> (12.0K)</a> |
 |
 |
14 |  | 
What is the correct name for this molecule?
 <a onClick="window.open('/olcweb/cgi/pluginpop.cgi?it=jpg::::/sites/dl/free/0072397462/209333/5_15_eq.jpg','popWin', 'width=NaN,height=NaN,resizable,scrollbars');" href="#"><img valign="absmiddle" height="16" width="16" border="0" src="/olcweb/styles/shared/linkicons/image.gif"> (15.0K)</a> <a onClick="window.open('/olcweb/cgi/pluginpop.cgi?it=jpg::::/sites/dl/free/0072397462/209333/5_15_eq.jpg','popWin', 'width=NaN,height=NaN,resizable,scrollbars');" href="#"><img valign="absmiddle" height="16" width="16" border="0" src="/olcweb/styles/shared/linkicons/image.gif"> (15.0K)</a> |
|  | A) | (2R,3R)-2-bromo-3-chlorobutane |
|  | B) | (2S,3R)-2-bromo-3-chlorobutane |
|  | C) | (2S,3S)-2-bromo-3-chlorobutane |
|  | D) | (2R,3S)-2-bromo-3-chlorobutane |
 |
 |
15 |  | 
The respective configurations of a and b are:
 <a onClick="window.open('/olcweb/cgi/pluginpop.cgi?it=jpg::::/sites/dl/free/0072397462/209333/5_16_eq.jpg','popWin', 'width=NaN,height=NaN,resizable,scrollbars');" href="#"><img valign="absmiddle" height="16" width="16" border="0" src="/olcweb/styles/shared/linkicons/image.gif"> (14.0K)</a> <a onClick="window.open('/olcweb/cgi/pluginpop.cgi?it=jpg::::/sites/dl/free/0072397462/209333/5_16_eq.jpg','popWin', 'width=NaN,height=NaN,resizable,scrollbars');" href="#"><img valign="absmiddle" height="16" width="16" border="0" src="/olcweb/styles/shared/linkicons/image.gif"> (14.0K)</a> |
|  | A) | (2R,3S) and (2R,3R) |
|  | B) | (2S,3R) and (2S,3R) |
|  | C) | (2S,3S) and (2R,3R) |
|  | D) | (2S,3R) and (2S,3S) |
 |
 |
16 |  | 
What is the specific rotation 20[α
]D of the following molecule?
 <a onClick="window.open('/olcweb/cgi/pluginpop.cgi?it=jpg::::/sites/dl/free/0072397462/209333/5_17_eq.jpg','popWin', 'width=NaN,height=NaN,resizable,scrollbars');" href="#"><img valign="absmiddle" height="16" width="16" border="0" src="/olcweb/styles/shared/linkicons/image.gif"> (18.0K)</a> <a onClick="window.open('/olcweb/cgi/pluginpop.cgi?it=jpg::::/sites/dl/free/0072397462/209333/5_17_eq.jpg','popWin', 'width=NaN,height=NaN,resizable,scrollbars');" href="#"><img valign="absmiddle" height="16" width="16" border="0" src="/olcweb/styles/shared/linkicons/image.gif"> (18.0K)</a> |
|  | A) | +4.42o |
|  | B) | +0.442o |
|  | C) | +44.2o |
|  | D) | – 44.2o |
 |
 |
17 |  | 
Which of the following physical properties differ for each of a pair of enantiomers? |
|  | A) | solubility in ethanol |
|  | B) | direction of rotation of plane-polarized light |
|  | C) | boiling point and melting point |
|  | D) | index of refraction |
 |
 |
18 |  | 
An optically active compound is composed of 75% of the (R) enantiomer and 25% of the (S) enantiomer. The enantiomeric excess (ee) is equal to |
|  | A) | 87.5%. |
|  | B) | 75%. |
|  | C) | 50%. |
|  | D) | 37.5%. |
 |
 |
19 |  | 
If a sample of 2-butanol has an enantiomeric excess of 60% of l-2-butanol, how much of each isomer is present? |
|  | A) | 60% levorotatory and 40% dextrorotatory |
|  | B) | 80% levorotatory and 20% dextrorotatory |
|  | C) | 70% levorotatory and 30% dextrorotatory |
|  | D) | 66% levorotatory and 34% dextrorotatory |
 |
 |
20 |  | 
How many stereocenters are there in the following molecule?
 <a onClick="window.open('/olcweb/cgi/pluginpop.cgi?it=jpg::::/sites/dl/free/0072397462/209333/5_20_eq.jpg','popWin', 'width=NaN,height=NaN,resizable,scrollbars');" href="#"><img valign="absmiddle" height="16" width="16" border="0" src="/olcweb/styles/shared/linkicons/image.gif"> (17.0K)</a> <a onClick="window.open('/olcweb/cgi/pluginpop.cgi?it=jpg::::/sites/dl/free/0072397462/209333/5_20_eq.jpg','popWin', 'width=NaN,height=NaN,resizable,scrollbars');" href="#"><img valign="absmiddle" height="16" width="16" border="0" src="/olcweb/styles/shared/linkicons/image.gif"> (17.0K)</a> |
|  | A) | 0 |
|  | B) | 2 |
|  | C) | 3 |
|  | D) | 1 |
 |
 |
21 |  | 
Which of the following statements is TRUE? |
|  | A) | To be diastereomers, a pair of molecules must have 2 or more chiral centers. |
|  | B) | To be diastereomers, a pair of molecules must have at least 1 chiral center. |
|  | C) | To be diastereomers, a pair of molecules must be stereoisomers. |
|  | D) | To be diastereomers, a pair of molecules must be a racemate. |
 |
 |
22 |  | 
Shikimic acid is an important natural product. How many stereoisomers are possible for shikimic acid?
 <a onClick="window.open('/olcweb/cgi/pluginpop.cgi?it=jpg::::/sites/dl/free/0072397462/209333/5_22_eq.jpg','popWin', 'width=NaN,height=NaN,resizable,scrollbars');" href="#"><img valign="absmiddle" height="16" width="16" border="0" src="/olcweb/styles/shared/linkicons/image.gif"> (15.0K)</a> <a onClick="window.open('/olcweb/cgi/pluginpop.cgi?it=jpg::::/sites/dl/free/0072397462/209333/5_22_eq.jpg','popWin', 'width=NaN,height=NaN,resizable,scrollbars');" href="#"><img valign="absmiddle" height="16" width="16" border="0" src="/olcweb/styles/shared/linkicons/image.gif"> (15.0K)</a> |
|  | A) | 16 |
|  | B) | 8 |
|  | C) | 4 |
|  | D) | 2 |
 |
 |
23 |  | 
Consider the following two structures. What is their isomeric relationship to each other?
 <a onClick="window.open('/olcweb/cgi/pluginpop.cgi?it=jpg::::/sites/dl/free/0072397462/209333/5_23_eq.jpg','popWin', 'width=NaN,height=NaN,resizable,scrollbars');" href="#"><img valign="absmiddle" height="16" width="16" border="0" src="/olcweb/styles/shared/linkicons/image.gif"> (16.0K)</a> <a onClick="window.open('/olcweb/cgi/pluginpop.cgi?it=jpg::::/sites/dl/free/0072397462/209333/5_23_eq.jpg','popWin', 'width=NaN,height=NaN,resizable,scrollbars');" href="#"><img valign="absmiddle" height="16" width="16" border="0" src="/olcweb/styles/shared/linkicons/image.gif"> (16.0K)</a> |
|  | A) | identical |
|  | B) | enantiomers |
|  | C) | diastereomers |
|  | D) | constitutional isomers |
 |
 |
24 |  | 
Explain why the two stereoisomers of 1,4-dimethylcyclohexane are achiral?
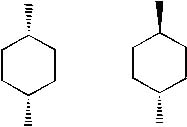 <a onClick="window.open('/olcweb/cgi/pluginpop.cgi?it=jpg::::/sites/dl/free/0072397462/209333/5_24_eq.jpg','popWin', 'width=NaN,height=NaN,resizable,scrollbars');" href="#"><img valign="absmiddle" height="16" width="16" border="0" src="/olcweb/styles/shared/linkicons/image.gif"> (14.0K)</a> <a onClick="window.open('/olcweb/cgi/pluginpop.cgi?it=jpg::::/sites/dl/free/0072397462/209333/5_24_eq.jpg','popWin', 'width=NaN,height=NaN,resizable,scrollbars');" href="#"><img valign="absmiddle" height="16" width="16" border="0" src="/olcweb/styles/shared/linkicons/image.gif"> (14.0K)</a> |
|  | A) | because they are identical |
|  | B) | because they are enantiomers |
|  | C) | because they both have the same absolute configuration |
|  | D) | because there are no stereocenters present in either molecule |
 |
 |
25 |  | 
How many stereoisomers are there for 1-ethyl-3-methylcyclohexane?
 <a onClick="window.open('/olcweb/cgi/pluginpop.cgi?it=jpg::::/sites/dl/free/0072397462/209333/5_25.jpg','popWin', 'width=NaN,height=NaN,resizable,scrollbars');" href="#"><img valign="absmiddle" height="16" width="16" border="0" src="/olcweb/styles/shared/linkicons/image.gif"> (12.0K)</a> <a onClick="window.open('/olcweb/cgi/pluginpop.cgi?it=jpg::::/sites/dl/free/0072397462/209333/5_25.jpg','popWin', 'width=NaN,height=NaN,resizable,scrollbars');" href="#"><img valign="absmiddle" height="16" width="16" border="0" src="/olcweb/styles/shared/linkicons/image.gif"> (12.0K)</a> |
|  | A) | 2 |
|  | B) | 3 |
|  | C) | 4 |
|  | D) | 6 |
 |
 |
26 |  | 
What is the relationship between (1R,2S)-dibromocyclohexane and (1S,2R)-dibromocyclohexane? |
|  | A) | identical |
|  | B) | enantiomers |
|  | C) | diastereomers |
|  | D) | constitutional isomers |
 |
 |
27 |  | 
What is the relationship between (1R,2S)-1-bromo-2-methylcyclohexane and (1S,2R)-1-bromo-2-methylcyclohexane? |
|  | A) | identical |
|  | B) | enantiomers |
|  | C) | diastereomers |
|  | D) | constitutional isomers |
 |
 |
28 |  | 
Which of the following may be separated by ordinary physical methods? |
|  | A) | a pair of identical molecules |
|  | B) | a pair of enantiomers |
|  | C) | a pair of diastereomers |
|  | D) | a pair of identical atoms |
 |
 |
29 |  | 
Which of the following may be separated by ordinary physical methods? |
|  | A) | (R)-3-bromo-1-butene and (S)-3-bromo-1-butene |
|  | B) | cis-2-bromo-2-butene and trans-2-bromo-2-butene |
|  | C) | (2R,3S)-1,2-dibromobutane and (2S,3R)-1,2-dibromobutane |
|  | D) | (R)-2-bromobutane and (S)-2-bromobutane |
 |
 |
30 |  | 
Consider the following two stereoisomers. How are they different?
 <a onClick="window.open('/olcweb/cgi/pluginpop.cgi?it=jpg::::/sites/dl/free/0072397462/209333/5_30_eq.jpg','popWin', 'width=NaN,height=NaN,resizable,scrollbars');" href="#"><img valign="absmiddle" height="16" width="16" border="0" src="/olcweb/styles/shared/linkicons/image.gif"> (13.0K)</a> <a onClick="window.open('/olcweb/cgi/pluginpop.cgi?it=jpg::::/sites/dl/free/0072397462/209333/5_30_eq.jpg','popWin', 'width=NaN,height=NaN,resizable,scrollbars');" href="#"><img valign="absmiddle" height="16" width="16" border="0" src="/olcweb/styles/shared/linkicons/image.gif"> (13.0K)</a> |
|  | A) | They have different melting points. |
|  | B) | They rotate plane-polarized light in opposite directions. |
|  | C) | They have different solubilities in water. |
|  | D) | They have different indices of refraction. |
 |

