 |
1 |  | 
Which set of steps is most typical of municipal drinking water purification in the U.S.? |
|  | A) | ozonation, distillation, chlorination |
|  | B) | flocculation, filtration, chlorination |
|  | C) | ultraviolet light, ion exchange, filtration |
|  | D) | distillation, chlorination, ozonation |
 |
 |
2 |  | 
Reverse osmosis, the process illustrated in this diagram, is a method used to
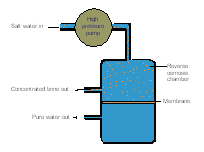 <a onClick="window.open('/olcweb/cgi/pluginpop.cgi?it=gif::::/sites/dl/free/0072410159/51619/5B2.gif','popWin', 'width=NaN,height=NaN,resizable,scrollbars');" href="#"><img valign="absmiddle" height="16" width="16" border="0" src="/olcweb/styles/shared/linkicons/image.gif"> (1.0K)</a> <a onClick="window.open('/olcweb/cgi/pluginpop.cgi?it=gif::::/sites/dl/free/0072410159/51619/5B2.gif','popWin', 'width=NaN,height=NaN,resizable,scrollbars');" href="#"><img valign="absmiddle" height="16" width="16" border="0" src="/olcweb/styles/shared/linkicons/image.gif"> (1.0K)</a>
|
|  | A) | produce bottled mineral water, particularly in Europe. |
|  | B) | rediscover dissolved gold from sea water. |
|  | C) | remove dissolved solids from salt water. |
|  | D) | purify municipal drinking water supplies in areas with soft water. |
 |
 |
3 |  | 
Which is a characteristic of a true solution? |
|  | A) | Only one solute can be present in a given amount of solvent. |
|  | B) | The solute and solvent are present in equal amounts. |
|  | C) | The solute cannot be separated from the solvent by simple filtration. |
|  | D) | The solute must be a solid and the solvent a liquid. |
 |
 |
4 |  | 
What is the molarity of a solution containing 3.5 moles of HCl in 5.0 L of solution? |
|  | A) | 0.70 M |
|  | B) | 1.4 M |
|  | C) | 3.5 M |
|  | D) | 18 M |
 |
 |
5 |  | 
Which statement is true for a calcium ion? |
|  | A) | number of protons = 20, number of electrons = 22, net charge = 2+ |
|  | B) | number of protons = 20, number of electrons = 18, net charge = 2- |
|  | C) | number of protons = 20, number of electrons = 22, net charge = 2- |
|  | D) | number of protons = 20, number of electrons = 18, net charge = 2+ |
 |
 |
6 |  | 
Which is the experimental H-to-O-to-H bond angle in a water molecule? |
|  | A) | 90o |
|  | B) | 104.5o |
|  | C) | 109.5o |
|  | D) | 180o |
 |
 |
7 |  | 
The gasoline additive MTBE has caused problems by contaminating ground water in some areas of the country where oxygenated fuels are required. What property of MTBE makes this problem worse?
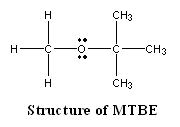 <a onClick="window.open('/olcweb/cgi/pluginpop.cgi?it=gif::::/sites/dl/free/0072410159/51619/5B7.gif','popWin', 'width=NaN,height=NaN,resizable,scrollbars');" href="#"><img valign="absmiddle" height="16" width="16" border="0" src="/olcweb/styles/shared/linkicons/image.gif"> (1.0K)</a> <a onClick="window.open('/olcweb/cgi/pluginpop.cgi?it=gif::::/sites/dl/free/0072410159/51619/5B7.gif','popWin', 'width=NaN,height=NaN,resizable,scrollbars');" href="#"><img valign="absmiddle" height="16" width="16" border="0" src="/olcweb/styles/shared/linkicons/image.gif"> (1.0K)</a>
|
|  | A) | MTBE is a nonpolar molecule and dissolves well in nonpolar soils, where it can leach into the ground water. |
|  | B) | MTBE is very volatile, and escapes into the air when gasoline is pumped. MTBE can then be washed into the soil by acid rain. |
|  | C) | MTBE is a polar molecule and, if oxygenated gasoline is spilled onto the ground, the MTBE dissolves into ground water. |
|  | D) | MTBE is not soluble in oxygenated gasolines, and therefore is never burned with the rest of the fuel. |
 |
 |
8 |  | 
Most of the earth's fresh water supply is found |
|  | A) | in the oceans. |
|  | B) | in plant material. |
|  | C) | as clouds in the atmosphere. |
|  | D) | in ice caps, glaciers, and ground water. |
 |
 |
9 |  | 
This diagram represents water molecules arranged in the liquid state. To what kind of bonding forces does the arrow point?
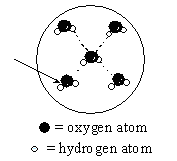 <a onClick="window.open('/olcweb/cgi/pluginpop.cgi?it=gif::::/sites/dl/free/0072410159/51619/5B9.gif','popWin', 'width=NaN,height=NaN,resizable,scrollbars');" href="#"><img valign="absmiddle" height="16" width="16" border="0" src="/olcweb/styles/shared/linkicons/image.gif"> (1.0K)</a> <a onClick="window.open('/olcweb/cgi/pluginpop.cgi?it=gif::::/sites/dl/free/0072410159/51619/5B9.gif','popWin', 'width=NaN,height=NaN,resizable,scrollbars');" href="#"><img valign="absmiddle" height="16" width="16" border="0" src="/olcweb/styles/shared/linkicons/image.gif"> (1.0K)</a>
|
|  | A) | a covalent bond, a type of intramolecular force |
|  | B) | a covalent bond, a type of intermolecular force |
|  | C) | a "hydrogen bond," a type of intramolecular force |
|  | D) | a "hydrogen bond," a type of intermolecular force |
 |
 |
10 |  | 
Which federal agency regulates the quality of bottled water in the U.S.? |
|  | A) | U.S. Food and Drug Administration, FDA |
|  | B) | U.S. Environmental Protection Agency, EPA |
|  | C) | U.S. Occupational Safety and Health Administration, OSHA |
|  | D) | U.S. Department of Agriculture, USDA |
 |
 |
11 |  | 
Numbers 1 - 4 are used to identify four different elements. Based on trends within the periodic table, which element is expected to have the greatest tendency to attract bonded electrons?
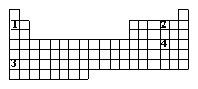 <a onClick="window.open('/olcweb/cgi/pluginpop.cgi?it=gif::::/sites/dl/free/0072410159/51619/5B11.gif','popWin', 'width=NaN,height=NaN,resizable,scrollbars');" href="#"><img valign="absmiddle" height="16" width="16" border="0" src="/olcweb/styles/shared/linkicons/image.gif"> (0.0K)</a> <a onClick="window.open('/olcweb/cgi/pluginpop.cgi?it=gif::::/sites/dl/free/0072410159/51619/5B11.gif','popWin', 'width=NaN,height=NaN,resizable,scrollbars');" href="#"><img valign="absmiddle" height="16" width="16" border="0" src="/olcweb/styles/shared/linkicons/image.gif"> (0.0K)</a>
|
|  | A) | element #1 |
|  | B) | element #2 |
|  | C) | element #3 |
|  | D) | element #4 |
 |
 |
12 |  | 
The MCLG for the nitrate ion in drinking water is 10 ppm. Express this value as a percentage. |
|  | A) | 1.0 x 105% |
|  | B) | 1.0 x 103% |
|  | C) | 1.0 x 10-2% |
|  | D) | 1.0 x 10-3% |
 |
 |
13 |  | 
Beer is a complex aqueous solution. For example, one brand has 7% by weight of ethanol. Ethanol has a formula weight of 46 g/mol. One liter of the 7% beer contains 62 g of ethanol. What is the approximate molarity of ethanol in this beer? |
|  | A) | 62 M |
|  | B) | 7 M |
|  | C) | 1.3 M |
|  | D) | 0.6 M |
 |
 |
14 |  | 
Which is true concerning chlorine molecules in water? |
|  | A) | Chlorine serves as an inexpensive, easily delivered, and persistent anti-bacterial in water. |
|  | B) | Chlorine reacts with small, naturally-occurring molecules in water to form halogenated organics. |
|  | C) | Chloroform and ozone are alternatives in current use as anti-microbial agents in water. |
|  | D) | Statements A and B are true. |
 |
 |
15 |  | 
Which mechanism(s) help explain how lead ions find their way into drinking water? |
|  | A) | residual lead (lead salts) from "leaded" gasoline that was burned during the 1950s, 1960s, and 1970s |
|  | B) | lead in pipes or connections between pipes |
|  | C) | lead leached from the soil as the result of chlorine in the water (formation of lead chloride salts) |
|  | D) | All of these are primary mechanisms for the delivery of lead into drinking water. |
 |

