 |
1 |  | 
The best sequence to use to form propylbenzene from benzene is |
|  | A) | Friedel–Crafts alkylation using 1-chloropropane and aluminum trichloride. |
|  | B) | Friedel–Crafts acylation followed by reduction. |
|  | C) | electrophilic addition of 1-chloropropane. |
|  | D) | nucleophilic aromatic substitution using propyl Grignard. |
 |
 |
2 |  | 
What is the electrophile in the electrophilic substitution reaction of benzene using HNO3 and H2SO4? |
|  | A) | SO3 |
|  | B) | NO3 |
|  | C) | NO2+ |
|  | D) | NO+ |
 |
 |
3 |  | 
How would you synthesize the following compound?
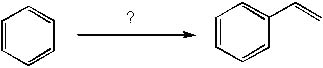 <a onClick="window.open('/olcweb/cgi/pluginpop.cgi?it=jpg::::/sites/dl/free/0072397462/209346/18_1.jpg','popWin', 'width=NaN,height=NaN,resizable,scrollbars');" href="#"><img valign="absmiddle" height="16" width="16" border="0" src="/olcweb/styles/shared/linkicons/image.gif"> (14.0K)</a> <a onClick="window.open('/olcweb/cgi/pluginpop.cgi?it=jpg::::/sites/dl/free/0072397462/209346/18_1.jpg','popWin', 'width=NaN,height=NaN,resizable,scrollbars');" href="#"><img valign="absmiddle" height="16" width="16" border="0" src="/olcweb/styles/shared/linkicons/image.gif"> (14.0K)</a> |
|  | A) | 1. MeCOCl, AlCl3
2. Zn (Hg) HCl |
|  | B) | 1. CH3COOH, AlCl3
2. H2, Pd
3. H2SO4, H2O, heat |
|  | C) | 1. CH3CH2Br, FeBr3
2. Br2, light
3. tBuONa, tBuOH, heat |
|  | D) | 1. CH3CHO, Mg in ether
2. NaBH4, MeOH
3. TsCl, pyridine
4. EtONa, EtOH
|
 |
 |
4 |  | 
The intermediate in the following reaction is a
 <a onClick="window.open('/olcweb/cgi/pluginpop.cgi?it=jpg::::/sites/dl/free/0072397462/209346/18_2.jpg','popWin', 'width=NaN,height=NaN,resizable,scrollbars');" href="#"><img valign="absmiddle" height="16" width="16" border="0" src="/olcweb/styles/shared/linkicons/image.gif"> (14.0K)</a> <a onClick="window.open('/olcweb/cgi/pluginpop.cgi?it=jpg::::/sites/dl/free/0072397462/209346/18_2.jpg','popWin', 'width=NaN,height=NaN,resizable,scrollbars');" href="#"><img valign="absmiddle" height="16" width="16" border="0" src="/olcweb/styles/shared/linkicons/image.gif"> (14.0K)</a> |
|  | A) | radical. |
|  | B) | carbanion. |
|  | C) | carbocation. |
|  | D) | transition state. |
 |
 |
5 |  | 
What is the electrophile in the electrophilic substitution reaction of acetyl chloride (CH3COCl) and AlCl3 reacting with benzene? |
|  | A) | Cl+ |
|  | B) | AlCl3 |
|  | C) | CH3C+=O |
|  | D) | C≡O+ |
 |
 |
6 |  | 
Which of the following aromatic compounds undergoes Friedel–Crafts alkylation with methyl chloride and aluminum chloride? |
|  | A) | benzoic acid |
|  | B) | nitrobenzene |
|  | C) | aniline |
|  | D) | toluene |
 |
 |
7 |  | 
Which of the following represents the best reagent(s) for the electrophilic nitration of benzene?
 <a onClick="window.open('/olcweb/cgi/pluginpop.cgi?it=jpg::::/sites/dl/free/0072397462/209346/18_7.jpg','popWin', 'width=NaN,height=NaN,resizable,scrollbars');" href="#"><img valign="absmiddle" height="16" width="16" border="0" src="/olcweb/styles/shared/linkicons/image.gif"> (13.0K)</a> <a onClick="window.open('/olcweb/cgi/pluginpop.cgi?it=jpg::::/sites/dl/free/0072397462/209346/18_7.jpg','popWin', 'width=NaN,height=NaN,resizable,scrollbars');" href="#"><img valign="absmiddle" height="16" width="16" border="0" src="/olcweb/styles/shared/linkicons/image.gif"> (13.0K)</a> |
|  | A) | NO2 |
|  | B) | HNO2 |
|  | C) | HNO3 |
|  | D) | H2SO4, HNO3 |
 |
 |
8 |  | 
Predict the major product of the following reaction.
 <a onClick="window.open('/olcweb/cgi/pluginpop.cgi?it=jpg::::/sites/dl/free/0072397462/209346/18_8.jpg','popWin', 'width=NaN,height=NaN,resizable,scrollbars');" href="#"><img valign="absmiddle" height="16" width="16" border="0" src="/olcweb/styles/shared/linkicons/image.gif"> (15.0K)</a> <a onClick="window.open('/olcweb/cgi/pluginpop.cgi?it=jpg::::/sites/dl/free/0072397462/209346/18_8.jpg','popWin', 'width=NaN,height=NaN,resizable,scrollbars');" href="#"><img valign="absmiddle" height="16" width="16" border="0" src="/olcweb/styles/shared/linkicons/image.gif"> (15.0K)</a> |
|  | A) | m-chlorophenol |
|  | B) | o-chlorophenol and p-chlorophenol |
|  | C) | o-hydroxytoluene and p-hydroxytoluene |
|  | D) | m-hydroxytoluene |
 |
 |
9 |  | 
Predict the major product of the following reaction.
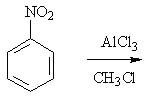 <a onClick="window.open('/olcweb/cgi/pluginpop.cgi?it=jpg::::/sites/dl/free/0072397462/209346/18_9.jpg','popWin', 'width=NaN,height=NaN,resizable,scrollbars');" href="#"><img valign="absmiddle" height="16" width="16" border="0" src="/olcweb/styles/shared/linkicons/image.gif"> (16.0K)</a> <a onClick="window.open('/olcweb/cgi/pluginpop.cgi?it=jpg::::/sites/dl/free/0072397462/209346/18_9.jpg','popWin', 'width=NaN,height=NaN,resizable,scrollbars');" href="#"><img valign="absmiddle" height="16" width="16" border="0" src="/olcweb/styles/shared/linkicons/image.gif"> (16.0K)</a> |
|  | A) | nitrobenzene |
|  | B) | o-nitrotoluene and p-nitrotoluene |
|  | C) | m-nitrotoluene |
|  | D) | chlorobenzene |
 |
 |
10 |  | 
Predict the major product of the following reaction.
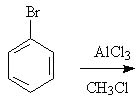 <a onClick="window.open('/olcweb/cgi/pluginpop.cgi?it=jpg::::/sites/dl/free/0072397462/209346/18_10.jpg','popWin', 'width=NaN,height=NaN,resizable,scrollbars');" href="#"><img valign="absmiddle" height="16" width="16" border="0" src="/olcweb/styles/shared/linkicons/image.gif"> (15.0K)</a> <a onClick="window.open('/olcweb/cgi/pluginpop.cgi?it=jpg::::/sites/dl/free/0072397462/209346/18_10.jpg','popWin', 'width=NaN,height=NaN,resizable,scrollbars');" href="#"><img valign="absmiddle" height="16" width="16" border="0" src="/olcweb/styles/shared/linkicons/image.gif"> (15.0K)</a> |
|  | A) | o-chlorobromobenzene and p-chlorobromobenzene |
|  | B) | m-chlorobromobenzene |
|  | C) | o-bromotoluene and p-bromotoluene |
|  | D) | m-bromotoluene |
 |
 |
11 |  | 
Predict the major product of the following reaction.
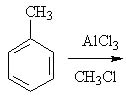 <a onClick="window.open('/olcweb/cgi/pluginpop.cgi?it=jpg::::/sites/dl/free/0072397462/209346/18_11.jpg','popWin', 'width=NaN,height=NaN,resizable,scrollbars');" href="#"><img valign="absmiddle" height="16" width="16" border="0" src="/olcweb/styles/shared/linkicons/image.gif"> (17.0K)</a> <a onClick="window.open('/olcweb/cgi/pluginpop.cgi?it=jpg::::/sites/dl/free/0072397462/209346/18_11.jpg','popWin', 'width=NaN,height=NaN,resizable,scrollbars');" href="#"><img valign="absmiddle" height="16" width="16" border="0" src="/olcweb/styles/shared/linkicons/image.gif"> (17.0K)</a> |
|  | A) | o-chlorotoluene and p-chlorotoluene |
|  | B) | m-chlorotoluene |
|  | C) | o-dimethylbenzene and p-dimethylbenzene |
|  | D) | m-dimethylbenzene |
 |
 |
12 |  | 
Predict the major product of the following reaction.
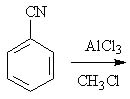 <a onClick="window.open('/olcweb/cgi/pluginpop.cgi?it=jpg::::/sites/dl/free/0072397462/209346/18_12.jpg','popWin', 'width=NaN,height=NaN,resizable,scrollbars');" href="#"><img valign="absmiddle" height="16" width="16" border="0" src="/olcweb/styles/shared/linkicons/image.gif"> (16.0K)</a> <a onClick="window.open('/olcweb/cgi/pluginpop.cgi?it=jpg::::/sites/dl/free/0072397462/209346/18_12.jpg','popWin', 'width=NaN,height=NaN,resizable,scrollbars');" href="#"><img valign="absmiddle" height="16" width="16" border="0" src="/olcweb/styles/shared/linkicons/image.gif"> (16.0K)</a> |
|  | A) | m-methylbenzenenitrile |
|  | B) | m-chlorobenzenenitrile |
|  | C) | o-cyanotoluene and p-cyanotoluene |
|  | D) | benzenenitrile |
 |
 |
13 |  | 
Predict the major product of the following reaction.
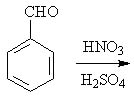 <a onClick="window.open('/olcweb/cgi/pluginpop.cgi?it=jpg::::/sites/dl/free/0072397462/209346/18_13.jpg','popWin', 'width=NaN,height=NaN,resizable,scrollbars');" href="#"><img valign="absmiddle" height="16" width="16" border="0" src="/olcweb/styles/shared/linkicons/image.gif"> (17.0K)</a> <a onClick="window.open('/olcweb/cgi/pluginpop.cgi?it=jpg::::/sites/dl/free/0072397462/209346/18_13.jpg','popWin', 'width=NaN,height=NaN,resizable,scrollbars');" href="#"><img valign="absmiddle" height="16" width="16" border="0" src="/olcweb/styles/shared/linkicons/image.gif"> (17.0K)</a> |
|  | A) | o-nitrobenzaldehyde and p-nitrobenzaldehyde |
|  | B) | m-nitrobenzaldehyde |
|  | C) | o-formylbenzenesulfonic acid and p-formylbenzenesulfonic acid |
|  | D) | m-formylbenzenesulfonic acid |
 |
 |
14 |  | 
Predict the major product of the following reaction.
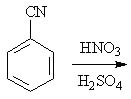 <a onClick="window.open('/olcweb/cgi/pluginpop.cgi?it=jpg::::/sites/dl/free/0072397462/209346/18_14.jpg','popWin', 'width=NaN,height=NaN,resizable,scrollbars');" href="#"><img valign="absmiddle" height="16" width="16" border="0" src="/olcweb/styles/shared/linkicons/image.gif"> (16.0K)</a> <a onClick="window.open('/olcweb/cgi/pluginpop.cgi?it=jpg::::/sites/dl/free/0072397462/209346/18_14.jpg','popWin', 'width=NaN,height=NaN,resizable,scrollbars');" href="#"><img valign="absmiddle" height="16" width="16" border="0" src="/olcweb/styles/shared/linkicons/image.gif"> (16.0K)</a> |
|  | A) | o-cyanonitrobenzene and p-cyanonitrobenzene |
|  | B) | m-cyanonitrobenzene |
|  | C) | o-cyanobenzenesulfonic acid and p-cyanobenzenesulfonic acid |
|  | D) | m-cyanobenzenesulfonic acid |
 |
 |
15 |  | 
Predict the major product of the following reaction.
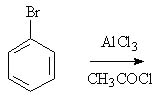 <a onClick="window.open('/olcweb/cgi/pluginpop.cgi?it=jpg::::/sites/dl/free/0072397462/209346/18_15.jpg','popWin', 'width=NaN,height=NaN,resizable,scrollbars');" href="#"><img valign="absmiddle" height="16" width="16" border="0" src="/olcweb/styles/shared/linkicons/image.gif"> (15.0K)</a> <a onClick="window.open('/olcweb/cgi/pluginpop.cgi?it=jpg::::/sites/dl/free/0072397462/209346/18_15.jpg','popWin', 'width=NaN,height=NaN,resizable,scrollbars');" href="#"><img valign="absmiddle" height="16" width="16" border="0" src="/olcweb/styles/shared/linkicons/image.gif"> (15.0K)</a> |
|  | A) | aceto-2-bromophenone and aceto-4-bromophenone |
|  | B) | aceto-(3-bromo)phenone |
|  | C) | o-bromochlorobenzene and p-bromochlorobenzene |
|  | D) | m-bromochlorobenzene |
 |
 |
16 |  | 
Predict the major product of the following reaction.
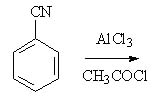 <a onClick="window.open('/olcweb/cgi/pluginpop.cgi?it=jpg::::/sites/dl/free/0072397462/209346/18_16.jpg','popWin', 'width=NaN,height=NaN,resizable,scrollbars');" href="#"><img valign="absmiddle" height="16" width="16" border="0" src="/olcweb/styles/shared/linkicons/image.gif"> (16.0K)</a> <a onClick="window.open('/olcweb/cgi/pluginpop.cgi?it=jpg::::/sites/dl/free/0072397462/209346/18_16.jpg','popWin', 'width=NaN,height=NaN,resizable,scrollbars');" href="#"><img valign="absmiddle" height="16" width="16" border="0" src="/olcweb/styles/shared/linkicons/image.gif"> (16.0K)</a> |
|  | A) | acetophenone |
|  | B) | benzoic acid |
|  | C) | benzamide |
|  | D) | cyanobenzene |
 |
 |
17 |  | 
Predict the major product of the following reaction.
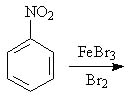 <a onClick="window.open('/olcweb/cgi/pluginpop.cgi?it=jpg::::/sites/dl/free/0072397462/209346/18_17.jpg','popWin', 'width=NaN,height=NaN,resizable,scrollbars');" href="#"><img valign="absmiddle" height="16" width="16" border="0" src="/olcweb/styles/shared/linkicons/image.gif"> (16.0K)</a> <a onClick="window.open('/olcweb/cgi/pluginpop.cgi?it=jpg::::/sites/dl/free/0072397462/209346/18_17.jpg','popWin', 'width=NaN,height=NaN,resizable,scrollbars');" href="#"><img valign="absmiddle" height="16" width="16" border="0" src="/olcweb/styles/shared/linkicons/image.gif"> (16.0K)</a> |
|  | A) | o-bromonitrobenzene and p-bromonitrobenzene |
|  | B) | m-bromonitrobenzene |
|  | C) | o-bromoaniline and p-bromoaniline |
|  | D) | m-bromoaniline |
 |
 |
18 |  | 
Predict the major product of the following reaction.
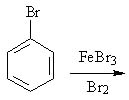 <a onClick="window.open('/olcweb/cgi/pluginpop.cgi?it=jpg::::/sites/dl/free/0072397462/209346/18_18.jpg','popWin', 'width=NaN,height=NaN,resizable,scrollbars');" href="#"><img valign="absmiddle" height="16" width="16" border="0" src="/olcweb/styles/shared/linkicons/image.gif"> (15.0K)</a> <a onClick="window.open('/olcweb/cgi/pluginpop.cgi?it=jpg::::/sites/dl/free/0072397462/209346/18_18.jpg','popWin', 'width=NaN,height=NaN,resizable,scrollbars');" href="#"><img valign="absmiddle" height="16" width="16" border="0" src="/olcweb/styles/shared/linkicons/image.gif"> (15.0K)</a> |
|  | A) | 1,2-dibromobenzene and 1,3-dibromobenzene |
|  | B) | 1,2-dibromobenzene and 1,4-dibromobenzene |
|  | C) | 1,3-dibromobenzene and 1,4-dibromobenzene |
|  | D) | 1,3-dibromobenzene |
 |
 |
19 |  | 
Predict the major product of the following reaction.
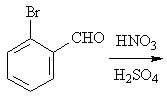 <a onClick="window.open('/olcweb/cgi/pluginpop.cgi?it=jpg::::/sites/dl/free/0072397462/209346/18_19.jpg','popWin', 'width=NaN,height=NaN,resizable,scrollbars');" href="#"><img valign="absmiddle" height="16" width="16" border="0" src="/olcweb/styles/shared/linkicons/image.gif"> (16.0K)</a> <a onClick="window.open('/olcweb/cgi/pluginpop.cgi?it=jpg::::/sites/dl/free/0072397462/209346/18_19.jpg','popWin', 'width=NaN,height=NaN,resizable,scrollbars');" href="#"><img valign="absmiddle" height="16" width="16" border="0" src="/olcweb/styles/shared/linkicons/image.gif"> (16.0K)</a> |
|  | A) | 2-bromo-6-nitrobenzaldehyde and 2-bromo-4-nitrobenzaldehyde |
|  | B) | 2-bromo-3-nitrobenzaldehyde and 2-bromo-5-nitrobenzaldehyde |
|  | C) | 2-bromo-5-nitrobenzaldehyde |
|  | D) | 2-bromo-3-nitrobenzaldehyde |
 |
 |
20 |  | 
Predict the major product of the following reaction.
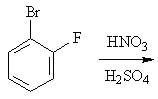 <a onClick="window.open('/olcweb/cgi/pluginpop.cgi?it=jpg::::/sites/dl/free/0072397462/209346/18_20.jpg','popWin', 'width=NaN,height=NaN,resizable,scrollbars');" href="#"><img valign="absmiddle" height="16" width="16" border="0" src="/olcweb/styles/shared/linkicons/image.gif"> (16.0K)</a> <a onClick="window.open('/olcweb/cgi/pluginpop.cgi?it=jpg::::/sites/dl/free/0072397462/209346/18_20.jpg','popWin', 'width=NaN,height=NaN,resizable,scrollbars');" href="#"><img valign="absmiddle" height="16" width="16" border="0" src="/olcweb/styles/shared/linkicons/image.gif"> (16.0K)</a> |
|  | A) | 1-bromo-2-fluoro-6-nitrobenzene and 1-bromo-2-fluoro-4-nitrobenzene |
|  | B) | 1-bromo-2-fluoro-3-nitrobenzene |
|  | C) | 1-bromo-2-fluoro-3-nitrobenzene and 1-bromo-2-fluoro-5-nitrobenzene |
|  | D) | 1-bromo-2-fluoro-6-nitrobenzene |
 |
 |
21 |  | 
Predict the major product of the following reaction.
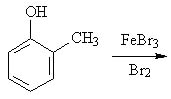 <a onClick="window.open('/olcweb/cgi/pluginpop.cgi?it=jpg::::/sites/dl/free/0072397462/209346/18_21.jpg','popWin', 'width=NaN,height=NaN,resizable,scrollbars');" href="#"><img valign="absmiddle" height="16" width="16" border="0" src="/olcweb/styles/shared/linkicons/image.gif"> (15.0K)</a> <a onClick="window.open('/olcweb/cgi/pluginpop.cgi?it=jpg::::/sites/dl/free/0072397462/209346/18_21.jpg','popWin', 'width=NaN,height=NaN,resizable,scrollbars');" href="#"><img valign="absmiddle" height="16" width="16" border="0" src="/olcweb/styles/shared/linkicons/image.gif"> (15.0K)</a> |
|  | A) | 2-bromo-6-methylphenol and 4-bromo-2-methylphenol |
|  | B) | 3-bromo-2-methylphenol and 5-bromo-2-methylphenol |
|  | C) | 5-bromo-2-methylphenol and 4-bromo-2-methylphenol |
|  | D) | 2-methylphenol |
 |
 |
22 |  | 
Predict the major product of the following reaction.
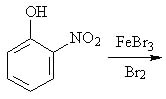 <a onClick="window.open('/olcweb/cgi/pluginpop.cgi?it=jpg::::/sites/dl/free/0072397462/209346/18_22.jpg','popWin', 'width=NaN,height=NaN,resizable,scrollbars');" href="#"><img valign="absmiddle" height="16" width="16" border="0" src="/olcweb/styles/shared/linkicons/image.gif"> (15.0K)</a> <a onClick="window.open('/olcweb/cgi/pluginpop.cgi?it=jpg::::/sites/dl/free/0072397462/209346/18_22.jpg','popWin', 'width=NaN,height=NaN,resizable,scrollbars');" href="#"><img valign="absmiddle" height="16" width="16" border="0" src="/olcweb/styles/shared/linkicons/image.gif"> (15.0K)</a> |
|  | A) | 3-bromo-2-nitrophenol and 5-bromo-2-nitrophenol |
|  | B) | 1-bromo-2-nitrobenzene |
|  | C) | 2-bromo-6-nitrophenol and 4-bromo-2-nitrophenol |
|  | D) | 2-hydroxyaniline |
 |
 |
23 |  | 
Which of the following is the most activating in electrophilic aromatic substitution?
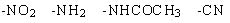 <a onClick="window.open('/olcweb/cgi/pluginpop.cgi?it=jpg::::/sites/dl/free/0072397462/209346/18_23.jpg','popWin', 'width=NaN,height=NaN,resizable,scrollbars');" href="#"><img valign="absmiddle" height="16" width="16" border="0" src="/olcweb/styles/shared/linkicons/image.gif"> (12.0K)</a> <a onClick="window.open('/olcweb/cgi/pluginpop.cgi?it=jpg::::/sites/dl/free/0072397462/209346/18_23.jpg','popWin', 'width=NaN,height=NaN,resizable,scrollbars');" href="#"><img valign="absmiddle" height="16" width="16" border="0" src="/olcweb/styles/shared/linkicons/image.gif"> (12.0K)</a> |
|  | A) | –NO2 |
|  | B) | –NH2 |
|  | C) | –NHCOCH3 |
|  | D) | –CN |
 |
 |
24 |  | 
Predict the major product(s) of the following reaction.
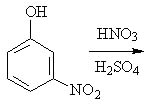 <a onClick="window.open('/olcweb/cgi/pluginpop.cgi?it=jpg::::/sites/dl/free/0072397462/209346/18_24.jpg','popWin', 'width=NaN,height=NaN,resizable,scrollbars');" href="#"><img valign="absmiddle" height="16" width="16" border="0" src="/olcweb/styles/shared/linkicons/image.gif"> (17.0K)</a> <a onClick="window.open('/olcweb/cgi/pluginpop.cgi?it=jpg::::/sites/dl/free/0072397462/209346/18_24.jpg','popWin', 'width=NaN,height=NaN,resizable,scrollbars');" href="#"><img valign="absmiddle" height="16" width="16" border="0" src="/olcweb/styles/shared/linkicons/image.gif"> (17.0K)</a> |
|  | A) | 3,5-dinitrophenol |
|  | B) | 2,3-dinitrophenol, 3,4-dinitrophenol, and 2,5-dinitrophenol |
|  | C) | 3,4-dinitrophenol, and 2,5-dinitrophenol |
|  | D) | 3,4-dinitrophenol |
 |
 |
25 |  | 
Predict the major product(s) of the following reaction.
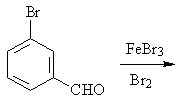 <a onClick="window.open('/olcweb/cgi/pluginpop.cgi?it=jpg::::/sites/dl/free/0072397462/209346/18_25.jpg','popWin', 'width=NaN,height=NaN,resizable,scrollbars');" href="#"><img valign="absmiddle" height="16" width="16" border="0" src="/olcweb/styles/shared/linkicons/image.gif"> (15.0K)</a> <a onClick="window.open('/olcweb/cgi/pluginpop.cgi?it=jpg::::/sites/dl/free/0072397462/209346/18_25.jpg','popWin', 'width=NaN,height=NaN,resizable,scrollbars');" href="#"><img valign="absmiddle" height="16" width="16" border="0" src="/olcweb/styles/shared/linkicons/image.gif"> (15.0K)</a> |
|  | A) | 3,5-dibromobenzaldehyde |
|  | B) | 2,3-dibromobenzaldehyde and 2,4-dibromobenzaldehyde and 2,5-dibromobenzaldehyde |
|  | C) | 2,3-dibromobenzaldehyde and 3,4-dibromobenzaldehyde and 2,5-dibromobenzaldehyde |
|  | D) | 3,4-dibromobenzaldehyde and 2,5-dibromobenzaldehyde |
 |
 |
26 |  | 
Predict the major product(s) of the following reaction.
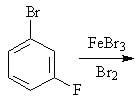 <a onClick="window.open('/olcweb/cgi/pluginpop.cgi?it=jpg::::/sites/dl/free/0072397462/209346/18_26.jpg','popWin', 'width=NaN,height=NaN,resizable,scrollbars');" href="#"><img valign="absmiddle" height="16" width="16" border="0" src="/olcweb/styles/shared/linkicons/image.gif"> (15.0K)</a> <a onClick="window.open('/olcweb/cgi/pluginpop.cgi?it=jpg::::/sites/dl/free/0072397462/209346/18_26.jpg','popWin', 'width=NaN,height=NaN,resizable,scrollbars');" href="#"><img valign="absmiddle" height="16" width="16" border="0" src="/olcweb/styles/shared/linkicons/image.gif"> (15.0K)</a> |
|  | A) | 1,3-dibromo-5-fluorobenzene |
|  | B) | 1,2-dibromo-4-fluorobenzene and 1,4-dibromo-2-fluorobenzene |
|  | C) | 1,2-dibromo-4-fluorobenzene and 1,4-dibromo-2-fluorobenzene and 1,2-dibromo-3-fluorobenzene |
|  | D) | 1,2-dibromo-3-fluorobenzene |
 |
 |
27 |  | 
Predict the major product of the following reaction.
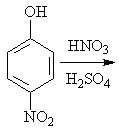 <a onClick="window.open('/olcweb/cgi/pluginpop.cgi?it=jpg::::/sites/dl/free/0072397462/209346/18_27.jpg','popWin', 'width=NaN,height=NaN,resizable,scrollbars');" href="#"><img valign="absmiddle" height="16" width="16" border="0" src="/olcweb/styles/shared/linkicons/image.gif"> (18.0K)</a> <a onClick="window.open('/olcweb/cgi/pluginpop.cgi?it=jpg::::/sites/dl/free/0072397462/209346/18_27.jpg','popWin', 'width=NaN,height=NaN,resizable,scrollbars');" href="#"><img valign="absmiddle" height="16" width="16" border="0" src="/olcweb/styles/shared/linkicons/image.gif"> (18.0K)</a> |
|  | A) | 2,4-dinitrophenol |
|  | B) | 3,4-dinitrophenol |
|  | C) | 2,4,6-trinitrophenol |
|  | D) | 3,4,5-trinitrophenol |
 |
 |
28 |  | 
Predict the major product of the following reaction.
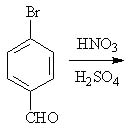 <a onClick="window.open('/olcweb/cgi/pluginpop.cgi?it=jpg::::/sites/dl/free/0072397462/209346/18_28.jpg','popWin', 'width=NaN,height=NaN,resizable,scrollbars');" href="#"><img valign="absmiddle" height="16" width="16" border="0" src="/olcweb/styles/shared/linkicons/image.gif"> (17.0K)</a> <a onClick="window.open('/olcweb/cgi/pluginpop.cgi?it=jpg::::/sites/dl/free/0072397462/209346/18_28.jpg','popWin', 'width=NaN,height=NaN,resizable,scrollbars');" href="#"><img valign="absmiddle" height="16" width="16" border="0" src="/olcweb/styles/shared/linkicons/image.gif"> (17.0K)</a> |
|  | A) | 4-bromo-2-nitrobenzaldehyde |
|  | B) | 4-bromo-3-nitrobenzaldehyde |
|  | C) | 4-bromo-2,6-dinitrobenzaldehyde |
|  | D) | 4-bromo-3,5-dinitrobenzaldehyde |
 |
 |
29 |  | 
Predict the major product of the following reaction.
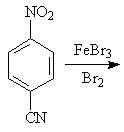 <a onClick="window.open('/olcweb/cgi/pluginpop.cgi?it=jpg::::/sites/dl/free/0072397462/209346/18_29.jpg','popWin', 'width=NaN,height=NaN,resizable,scrollbars');" href="#"><img valign="absmiddle" height="16" width="16" border="0" src="/olcweb/styles/shared/linkicons/image.gif"> (17.0K)</a> <a onClick="window.open('/olcweb/cgi/pluginpop.cgi?it=jpg::::/sites/dl/free/0072397462/209346/18_29.jpg','popWin', 'width=NaN,height=NaN,resizable,scrollbars');" href="#"><img valign="absmiddle" height="16" width="16" border="0" src="/olcweb/styles/shared/linkicons/image.gif"> (17.0K)</a> |
|  | A) | 2,6-dibromo-4-nitrobenzonitrile |
|  | B) | 3,5-dibromo-4-nitrobenzonitrile |
|  | C) | 2-bromo-4-nitrobenzonitrile |
|  | D) | 3-bromo-4-nitrobenzonitrile |
 |
 |
30 |  | 
Predict the major product of the following reaction.
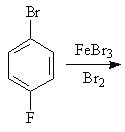 <a onClick="window.open('/olcweb/cgi/pluginpop.cgi?it=jpg::::/sites/dl/free/0072397462/209346/18_30.jpg','popWin', 'width=NaN,height=NaN,resizable,scrollbars');" href="#"><img valign="absmiddle" height="16" width="16" border="0" src="/olcweb/styles/shared/linkicons/image.gif"> (15.0K)</a> <a onClick="window.open('/olcweb/cgi/pluginpop.cgi?it=jpg::::/sites/dl/free/0072397462/209346/18_30.jpg','popWin', 'width=NaN,height=NaN,resizable,scrollbars');" href="#"><img valign="absmiddle" height="16" width="16" border="0" src="/olcweb/styles/shared/linkicons/image.gif"> (15.0K)</a> |
|  | A) | 1,2-dibromo-4-fluorobenzene |
|  | B) | 1,2,6-tribromo-4-fluorobenzene |
|  | C) | 1,3-dibromo-4-fluorobenzene |
|  | D) | 1,3,5-tribromo-4-fluorobenzene |
 |

