Please note that this quiz uses special characters. Not all browsers display special characters properly. For best results please use Internet Explorer 5.5 or newer or Netscape 6.2 or newer.
 |
1 |  | 
The lone pair of electrons on sulfur in SO2 is in
|
|  | A) | a p orbital.
|
|  | B) | an s orbital.
|
|  | C) | an sp2 hybrid orbital.
|
|  | D) | an sp3 hybrid orbital.
|
|  | E) | none of the above orbital types.
|
 |
 |
2 |  | 
Which of the following molecules has sp3 hybridization and a dipole moment?
|
|  | A) | SiH4 |
|  | B) | BF3 |
|  | C) | NH3 |
|  | D) | BrF3 |
|  | E) | PCl5 |
 |
 |
3 |  | 
Which of the following hybridization patterns is associated with trigonal bipyramidal geometry?
|
|  | A) | sp2 |
|  | B) | sp3 |
|  | C) | sp3d2 |
|  | D) | sp3d |
|  | E) | sp |
 |
 |
4 |  | 
Which of the following hybridization patterns best describes the central atom in H2CF2?
|
|  | A) | sp2 |
|  | B) | sp3d |
|  | C) | sp3 |
|  | D) | sp |
|  | E) | sp3d2 |
 |
 |
5 |  | 
How many 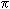 <a onClick="window.open('/olcweb/cgi/pluginpop.cgi?it=gif:: ::/sites/dl/free/0072396814/37547/pi.gif','popWin', 'width=NaN,height=NaN,resizable,scrollbars');" href="#"><img valign="absmiddle" height="16" width="16" border="0" src="/olcweb/styles/shared/linkicons/image.gif"> (0.0K)</a> bonds are present in a molecule of ethyne (acetylene), C2H2? <a onClick="window.open('/olcweb/cgi/pluginpop.cgi?it=gif:: ::/sites/dl/free/0072396814/37547/pi.gif','popWin', 'width=NaN,height=NaN,resizable,scrollbars');" href="#"><img valign="absmiddle" height="16" width="16" border="0" src="/olcweb/styles/shared/linkicons/image.gif"> (0.0K)</a> bonds are present in a molecule of ethyne (acetylene), C2H2? |
|  | A) | 3 |
|  | B) | 2 |
|  | C) | 1 |
|  | D) | 0 |
|  | E) | none of the above |
 |
 |
6 |  | 
Which of the following hybridization patterns best describes the carbon atom in NCO-? |
|  | A) | sp2 |
|  | B) | sp3d |
|  | C) | sp3 |
|  | D) | sp |
|  | E) | sp3d2 |
 |
 |
7 |  | 
A carbon-carbon double bond consists of |
|  | A) | a  <a onClick="window.open('/olcweb/cgi/pluginpop.cgi?it=gif:: ::/sites/dl/free/0072396814/37547/sigma.gif','popWin', 'width=NaN,height=NaN,resizable,scrollbars');" href="#"><img valign="absmiddle" height="16" width="16" border="0" src="/olcweb/styles/shared/linkicons/image.gif"> (0.0K)</a> bond and two <a onClick="window.open('/olcweb/cgi/pluginpop.cgi?it=gif:: ::/sites/dl/free/0072396814/37547/sigma.gif','popWin', 'width=NaN,height=NaN,resizable,scrollbars');" href="#"><img valign="absmiddle" height="16" width="16" border="0" src="/olcweb/styles/shared/linkicons/image.gif"> (0.0K)</a> bond and two 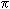 <a onClick="window.open('/olcweb/cgi/pluginpop.cgi?it=gif:: ::/sites/dl/free/0072396814/37547/pi.gif','popWin', 'width=NaN,height=NaN,resizable,scrollbars');" href="#"><img valign="absmiddle" height="16" width="16" border="0" src="/olcweb/styles/shared/linkicons/image.gif"> (0.0K)</a> bonds. <a onClick="window.open('/olcweb/cgi/pluginpop.cgi?it=gif:: ::/sites/dl/free/0072396814/37547/pi.gif','popWin', 'width=NaN,height=NaN,resizable,scrollbars');" href="#"><img valign="absmiddle" height="16" width="16" border="0" src="/olcweb/styles/shared/linkicons/image.gif"> (0.0K)</a> bonds. |
|  | B) | a 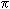 <a onClick="window.open('/olcweb/cgi/pluginpop.cgi?it=gif:: ::/sites/dl/free/0072396814/37547/pi.gif','popWin', 'width=NaN,height=NaN,resizable,scrollbars');" href="#"><img valign="absmiddle" height="16" width="16" border="0" src="/olcweb/styles/shared/linkicons/image.gif"> (0.0K)</a> bond and two <a onClick="window.open('/olcweb/cgi/pluginpop.cgi?it=gif:: ::/sites/dl/free/0072396814/37547/pi.gif','popWin', 'width=NaN,height=NaN,resizable,scrollbars');" href="#"><img valign="absmiddle" height="16" width="16" border="0" src="/olcweb/styles/shared/linkicons/image.gif"> (0.0K)</a> bond and two  <a onClick="window.open('/olcweb/cgi/pluginpop.cgi?it=gif:: ::/sites/dl/free/0072396814/37547/sigma.gif','popWin', 'width=NaN,height=NaN,resizable,scrollbars');" href="#"><img valign="absmiddle" height="16" width="16" border="0" src="/olcweb/styles/shared/linkicons/image.gif"> (0.0K)</a> bonds. <a onClick="window.open('/olcweb/cgi/pluginpop.cgi?it=gif:: ::/sites/dl/free/0072396814/37547/sigma.gif','popWin', 'width=NaN,height=NaN,resizable,scrollbars');" href="#"><img valign="absmiddle" height="16" width="16" border="0" src="/olcweb/styles/shared/linkicons/image.gif"> (0.0K)</a> bonds. |
|  | C) | two  <a onClick="window.open('/olcweb/cgi/pluginpop.cgi?it=gif:: ::/sites/dl/free/0072396814/37547/sigma.gif','popWin', 'width=NaN,height=NaN,resizable,scrollbars');" href="#"><img valign="absmiddle" height="16" width="16" border="0" src="/olcweb/styles/shared/linkicons/image.gif"> (0.0K)</a> bonds. <a onClick="window.open('/olcweb/cgi/pluginpop.cgi?it=gif:: ::/sites/dl/free/0072396814/37547/sigma.gif','popWin', 'width=NaN,height=NaN,resizable,scrollbars');" href="#"><img valign="absmiddle" height="16" width="16" border="0" src="/olcweb/styles/shared/linkicons/image.gif"> (0.0K)</a> bonds. |
|  | D) | two 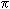 <a onClick="window.open('/olcweb/cgi/pluginpop.cgi?it=gif:: ::/sites/dl/free/0072396814/37547/pi.gif','popWin', 'width=NaN,height=NaN,resizable,scrollbars');" href="#"><img valign="absmiddle" height="16" width="16" border="0" src="/olcweb/styles/shared/linkicons/image.gif"> (0.0K)</a> bonds. <a onClick="window.open('/olcweb/cgi/pluginpop.cgi?it=gif:: ::/sites/dl/free/0072396814/37547/pi.gif','popWin', 'width=NaN,height=NaN,resizable,scrollbars');" href="#"><img valign="absmiddle" height="16" width="16" border="0" src="/olcweb/styles/shared/linkicons/image.gif"> (0.0K)</a> bonds. |
|  | E) | a  <a onClick="window.open('/olcweb/cgi/pluginpop.cgi?it=gif:: ::/sites/dl/free/0072396814/37547/sigma.gif','popWin', 'width=NaN,height=NaN,resizable,scrollbars');" href="#"><img valign="absmiddle" height="16" width="16" border="0" src="/olcweb/styles/shared/linkicons/image.gif"> (0.0K)</a> bond and a <a onClick="window.open('/olcweb/cgi/pluginpop.cgi?it=gif:: ::/sites/dl/free/0072396814/37547/sigma.gif','popWin', 'width=NaN,height=NaN,resizable,scrollbars');" href="#"><img valign="absmiddle" height="16" width="16" border="0" src="/olcweb/styles/shared/linkicons/image.gif"> (0.0K)</a> bond and a 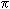 <a onClick="window.open('/olcweb/cgi/pluginpop.cgi?it=gif:: ::/sites/dl/free/0072396814/37547/pi.gif','popWin', 'width=NaN,height=NaN,resizable,scrollbars');" href="#"><img valign="absmiddle" height="16" width="16" border="0" src="/olcweb/styles/shared/linkicons/image.gif"> (0.0K)</a> bond. <a onClick="window.open('/olcweb/cgi/pluginpop.cgi?it=gif:: ::/sites/dl/free/0072396814/37547/pi.gif','popWin', 'width=NaN,height=NaN,resizable,scrollbars');" href="#"><img valign="absmiddle" height="16" width="16" border="0" src="/olcweb/styles/shared/linkicons/image.gif"> (0.0K)</a> bond. |
 |
 |
8 |  | 
What type orbital holds the lone pair of electrons on nitrogen in H2CNH?
|
|  | A) | 2s |
|  | B) | 2px |
|  | C) | sp |
|  | D) | sp2 |
|  | E) | sp3 |
 |
 |
9 |  | 
In a carbon-carbon triple bond, what is the nature of the bonding between the carbons?
|
|  | A) | two 2s orbitals overlapping
|
|  | B) | two 2p orbitals overlapping
|
|  | C) | two sp orbitals overlapping, two 2py overlapping and two 2pz overlapping
|
|  | D) | an sp and sp2 overlapping and 2p orbitals overlapping
|
|  | E) | an sp2 and sp2 overlapping and 2p orbitals overlapping
|
 |
 |
10 |  | 
Which type of hybrid orbital on carbon is associated with bonds having restricted rotation?
|
|  | A) | sp |
|  | B) | sp2 |
|  | C) | sp3 |
|  | D) | sp3d |
|  | E) | sp3d2 |
 |
 |
11 |  | 
When the nitrogen molecule, N2, loses an electron,
|
|  | A) | the ion formed will be less stable than N2.
|
|  | B) | the ion formed will be more stable than N2.
|
|  | C) | the stability of the ion formed will be the same as that of N2.
|
|  | D) | the bond length of ion will be shorter than the bond length of N2.
|
|  | E) | none of the above statements is correct.
|
 |
 |
12 |  | 
According to the molecular orbital model, which is the least stable?
|
|  | A) | He2 |
|  | B) | Li2 |
|  | C) | B2 |
|  | D) | C2 |
|  | E) | NO
|
 |
 |
13 |  | 
According to molecular orbital theory, what is the bond order in F2+?
|
|  | A) | 1
|
|  | B) | 1.5
|
|  | C) | 2
|
|  | D) | 2.5
|
|  | E) | 3.0
|
 |
 |
14 |  | 
According to the molecular orbital model, the highest occupied molecular orbital of O2 is |
|  | A) |  <a onClick="window.open('/olcweb/cgi/pluginpop.cgi?it=gif:: ::/sites/dl/free/0072396814/37547/sigma.gif','popWin', 'width=NaN,height=NaN,resizable,scrollbars');" href="#"><img valign="absmiddle" height="16" width="16" border="0" src="/olcweb/styles/shared/linkicons/image.gif"> (0.0K)</a> 2s <a onClick="window.open('/olcweb/cgi/pluginpop.cgi?it=gif:: ::/sites/dl/free/0072396814/37547/sigma.gif','popWin', 'width=NaN,height=NaN,resizable,scrollbars');" href="#"><img valign="absmiddle" height="16" width="16" border="0" src="/olcweb/styles/shared/linkicons/image.gif"> (0.0K)</a> 2s |
|  | B) | 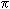 <a onClick="window.open('/olcweb/cgi/pluginpop.cgi?it=gif:: ::/sites/dl/free/0072396814/37547/pi.gif','popWin', 'width=NaN,height=NaN,resizable,scrollbars');" href="#"><img valign="absmiddle" height="16" width="16" border="0" src="/olcweb/styles/shared/linkicons/image.gif"> (0.0K)</a> 2p <a onClick="window.open('/olcweb/cgi/pluginpop.cgi?it=gif:: ::/sites/dl/free/0072396814/37547/pi.gif','popWin', 'width=NaN,height=NaN,resizable,scrollbars');" href="#"><img valign="absmiddle" height="16" width="16" border="0" src="/olcweb/styles/shared/linkicons/image.gif"> (0.0K)</a> 2p |
|  | C) |  <a onClick="window.open('/olcweb/cgi/pluginpop.cgi?it=gif:: ::/sites/dl/free/0072396814/37547/sigma.gif','popWin', 'width=NaN,height=NaN,resizable,scrollbars');" href="#"><img valign="absmiddle" height="16" width="16" border="0" src="/olcweb/styles/shared/linkicons/image.gif"> (0.0K)</a> *2s <a onClick="window.open('/olcweb/cgi/pluginpop.cgi?it=gif:: ::/sites/dl/free/0072396814/37547/sigma.gif','popWin', 'width=NaN,height=NaN,resizable,scrollbars');" href="#"><img valign="absmiddle" height="16" width="16" border="0" src="/olcweb/styles/shared/linkicons/image.gif"> (0.0K)</a> *2s |
|  | D) | 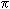 <a onClick="window.open('/olcweb/cgi/pluginpop.cgi?it=gif:: ::/sites/dl/free/0072396814/37547/pi.gif','popWin', 'width=NaN,height=NaN,resizable,scrollbars');" href="#"><img valign="absmiddle" height="16" width="16" border="0" src="/olcweb/styles/shared/linkicons/image.gif"> (0.0K)</a> *2p <a onClick="window.open('/olcweb/cgi/pluginpop.cgi?it=gif:: ::/sites/dl/free/0072396814/37547/pi.gif','popWin', 'width=NaN,height=NaN,resizable,scrollbars');" href="#"><img valign="absmiddle" height="16" width="16" border="0" src="/olcweb/styles/shared/linkicons/image.gif"> (0.0K)</a> *2p |
|  | E) |  <a onClick="window.open('/olcweb/cgi/pluginpop.cgi?it=gif:: ::/sites/dl/free/0072396814/37547/sigma.gif','popWin', 'width=NaN,height=NaN,resizable,scrollbars');" href="#"><img valign="absmiddle" height="16" width="16" border="0" src="/olcweb/styles/shared/linkicons/image.gif"> (0.0K)</a> *2p <a onClick="window.open('/olcweb/cgi/pluginpop.cgi?it=gif:: ::/sites/dl/free/0072396814/37547/sigma.gif','popWin', 'width=NaN,height=NaN,resizable,scrollbars');" href="#"><img valign="absmiddle" height="16" width="16" border="0" src="/olcweb/styles/shared/linkicons/image.gif"> (0.0K)</a> *2p |
 |
 |
15 |  | 
Consider benzene (C6H6), carbonate ion (CO32-) and water (H2O). Of these three species, which
one(s) has/have delocalized molecular orbitals?
|
|  | A) | only benzene
|
|  | B) | only carbonate
|
|  | C) | only water
|
|  | D) | benzene and water
|
|  | E) | benzene and carbonate
|
 |

