 |
| 1 |  | 
Iron has an atomic number of 26 and a mass number of 56. How many protons, neutrons, and electrons are in an atom of iron? If an atom of iron lost three electrons, what would the charge of the resulting ion be? Write the correct symbol for this ion. |
|  | |
|
|
 |
| 2 |  | 
Which of the following pairs of terms applies to the reaction that results in the formation of fatty acids and glycerol from a triglyceride molecule?
- Decomposition or synthesis reaction
- Anabolism or catabolism
- Dehydration or hydrolysis reaction
|
|  | |
|
|
 |
| 3 |  | 
A mixture of chemicals is warmed slightly. As a consequence, although no more heat is added, the solution becomes very hot. Explain what occurred to make the solution so hot. |
|  | |
|
|
 |
| 4 |  | 
Two solutions, when mixed together at room temperature, produce a chemical reaction. When the solutions are boiled and allowed to cool to room temperature before mixing, however, no chemical reaction takes place. Explain. |
|  | |
|
|
 |
| 5 |  | 
In terms of the potential energy in the food, explain why eating food is necessary for increasing muscle mass. |
|  | |
|
|
 |
| 6 |  | 
Solution A has a pH of 2, and solution B has a pH of 8. If equal amounts of solutions A and B are mixed, is the resulting solution acidic or basic? |
|  | |
|
|
 |
| 7 |  | 
Given a buffered solution that is based on the following equilibrium:
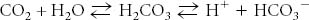 <a onClick="window.open('/olcweb/cgi/pluginpop.cgi?it=jpg::::/sites/dl/free/0072507470/234416/eq_ch02_q7_eqn01.jpg','popWin', 'width=NaN,height=NaN,resizable,scrollbars');" href="#"><img valign="absmiddle" height="16" width="16" border="0" src="/olcweb/styles/shared/linkicons/image.gif"> (11.0K)</a> <a onClick="window.open('/olcweb/cgi/pluginpop.cgi?it=jpg::::/sites/dl/free/0072507470/234416/eq_ch02_q7_eqn01.jpg','popWin', 'width=NaN,height=NaN,resizable,scrollbars');" href="#"><img valign="absmiddle" height="16" width="16" border="0" src="/olcweb/styles/shared/linkicons/image.gif"> (11.0K)</a>
what happens to the pH of the solution if NaHCO3 is added? |
|  | |
|
|
 |
| 8 |  | 
An enzyme E catalyzes the following reaction:
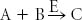 <a onClick="window.open('/olcweb/cgi/pluginpop.cgi?it=jpg::::/sites/dl/free/0072507470/234416/eq_ch02_q8_eqn01.jpg','popWin', 'width=NaN,height=NaN,resizable,scrollbars');" href="#"><img valign="absmiddle" height="16" width="16" border="0" src="/olcweb/styles/shared/linkicons/image.gif"> (9.0K)</a> <a onClick="window.open('/olcweb/cgi/pluginpop.cgi?it=jpg::::/sites/dl/free/0072507470/234416/eq_ch02_q8_eqn01.jpg','popWin', 'width=NaN,height=NaN,resizable,scrollbars');" href="#"><img valign="absmiddle" height="16" width="16" border="0" src="/olcweb/styles/shared/linkicons/image.gif"> (9.0K)</a>
The product C, however, binds to the active site of the enzyme in a reversible fashion and keeps the enzyme from functioning. What happens if A and B are continually added to a solution that contains a fixed amount of the enzyme? |
|  | |
|
|
 |
| 9 |  | 
Given the materials commonly found in a kitchen, explain how one could distinguish between a protein and a lipid. |
|  | |
|
|
 |
| 10 |  | 
A student is given two unlabeled substances: one a typical phospholipid and one a typical protein. She is asked to determine which substance is the protein and which is the phospholipid. The available techniques allow her to determine the elements in each sample. How can she identify each substance? |
|  | |
|
|

